More info
|
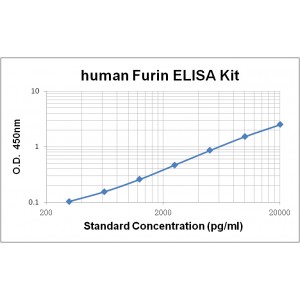
Assay Range |
312 -20,000 pg/mL |
|
Sensitivity |
10.0 pg/mL |
|
Size |
96T |
|
Storage |
Store at 2 - 8ºC. Keep reconstituted standard and detection Ab at -20 ºC |
|
Assay Principle |
Sandwich ELISA |
|
Sample Volume |
100 µL final volume, dilution factor varies on samples |
|
Detection Method |
Chromogenic |
Kit Components
1. Recombinant Human Furin standard: 2 vials
2. One 96-well plate coated with Human Furin Ab
3. Sample diluent buffer: 12 mL - 1
4. Detection antibody: 130 µL, dilution 1:100
5. Streptavidin-HRP: 130 µL, dilution 1:100
6. Antibody diluent buffer: 12 mL x1
7. Streptavidin-HRP diluent buffer: 12 mL x1
8. TMB developing agent: 10 mL x1
9. Stop solution: 10 mL x1
10. Washing solution (20x): 25 mL x1
Background
Furin, also known as dibasic-processing enzyme, paired basic amino acid residue-cleaving enzyme (PACE), is a member of the proprotein convertase (PC) family, which belongs to the subtilisin superfamily of serine protease. Furin is synthesized as a 794 amino acid (aa) type I transmembrane protein precursor with a 124 aa signal peptide, a 83 aa pro region, which play a crucial role in the folding, activation and transport of Furin, and a 587 aa mature chain. The mature chain consists of the subtilisin-like catalytic domain, a P domain, which is essential for enzyme activity and the modulation of pH and calcium requirements, and a cytoplasmic domain, which controls the localization and sorting of Furin in the trans-Golgi network/endosomal system. Furin processes a variety of proproteins in compartments of secretory pathways by cleaving after ArgXaaLys/ArgArg like motifs, which usually reside at the end of the pro regions of these proproteins. Furin is enriched in the Golgi apparatus, where it cleaves the proproteins into their mature/active forms. Furin plays an essential role in embryogenesis and homeostasis and is implicated in various pathologies such as cancer, neurodegenerative diseases and anthrax. It is reported that a furin-like pro-protein convertase has been implicated in the processing of RGMc/hemojuvelin, a protein involved in a severe iron-overload disorder called juvenile hemochromatosis. In addition to processing cellular precursor proteins, furin is also utilized by a number of pathogens. For example, the envelope proteins of viruses such as HIV, influenza and dengue fever viruses must be cleaved by furin or furin-like proteases to become fully functional. Anthrax toxin, pseudomonas exotoxin, and papillomaviruses must be processed by furin during their initial entry into host cells.